|
If the second element is oxygen, the trailing vowel is usually omitted from the end of a polysyllabic prefix but not a monosyllabic one (that is, we would say “monoxide” rather than “monooxide” and “trioxide” rather than “troxide”). Normally, no prefix is added to the first element’s name if there is only one atom of the first element in a molecule. Table 4.1 “Numerical Prefixes for Naming Binary Covalent Compounds” lists these numerical prefixes. CCl4 (carbon tetrachloride) is a covalent compound. The second element, chlorine, becomes chloride, and we attach the correct numerical prefix (tetra-) to indicate that the molecule contains four chlorine atoms. Properties of Ionic Compounds The crystal structure of ionic compounds is. A system of numerical prefixes is used to specify the number of atoms in a molecule. Let us practice by naming the compound whose molecular formula is CCl 4.The name begins with the name of the first elementcarbon. The second element is named by taking the stem of the element name and adding the suffix – ide. Putting these pieces together gives the name carbon tetrachloride for this compound. The elements in (N2O4) are both nonmetals, rather than a metal and a nonmetal. so this compound contains both ionic and covalent bonds. The first element in the formula is simply listed using the name of the element. The chemical formula of a simple covalent compound can be determined from its name. 1: Sharing is caring, especially for atoms that participate in covalent bonding. In this class, we will not discuss the option of metallic bonding which is a form of covalent bonding. Two combinations of atoms can produce this type of bonding: nonmetal/nonmetal or metalloid/nonmetal. Naming binary (two-element) covalent compounds is similar to naming simple ionic compounds. This type of bonding would be a covalent bond. 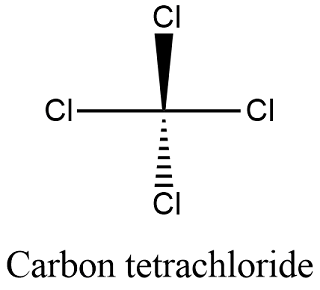
For example, we have already seen CH 4, the molecular formula for methane. Numerical subscripts are used if there is more than one of a particular atom. Carbon disulfide (also spelled as carbon disulphide) is an inorganic compound with the chemical formula CS 2 and structure SCS.It is a colorless, flammable, neurotoxic liquid that is used as a building block in organic synthesis. Then the other nonmetal symbols are listed. Typically, a molecular formula begins with the nonmetal that is closest to the lower left corner of the periodic table, except that hydrogen is almost never written first (H 2O is the prominent exception). The other fluoride of tin is SnF 4, which was previously called stannic fluoride but is now named tin(IV) fluoride.Ĭhemists use nomenclature rules to clearly name compounds.The chemical formulas for covalent compounds are referred to as molecular formulas because these compounds exist as separate, discrete molecules. This represents the formula SnF 2, which is more properly named tin(II) fluoride. CCl 4 is a clear, nonflammable, heavy liquid that evaporates readily, producing a sweet characteristic odor similar to chloroform. Its chemical structure is shown below: Figure 1. For example, you may see the words stannous fluoride on a tube of toothpaste. Carbon tetrachloride (CCl 4) is a manufactured chemical that does not occur naturally in the environment. Though this naming convention has been largely abandoned by the scientific community, it remains in use by some segments of industry. Out-of-date nomenclature used the suffixes – ic and – ous to designate metals with higher and lower charges, respectively: Iron(III) chloride, FeCl 3, was previously called ferric chloride, and iron(II) chloride, FeCl 2, was known as ferrous chloride. \): Names of Some Transition Metal Ionic Compounds Transition Metal Ionic Compound
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
